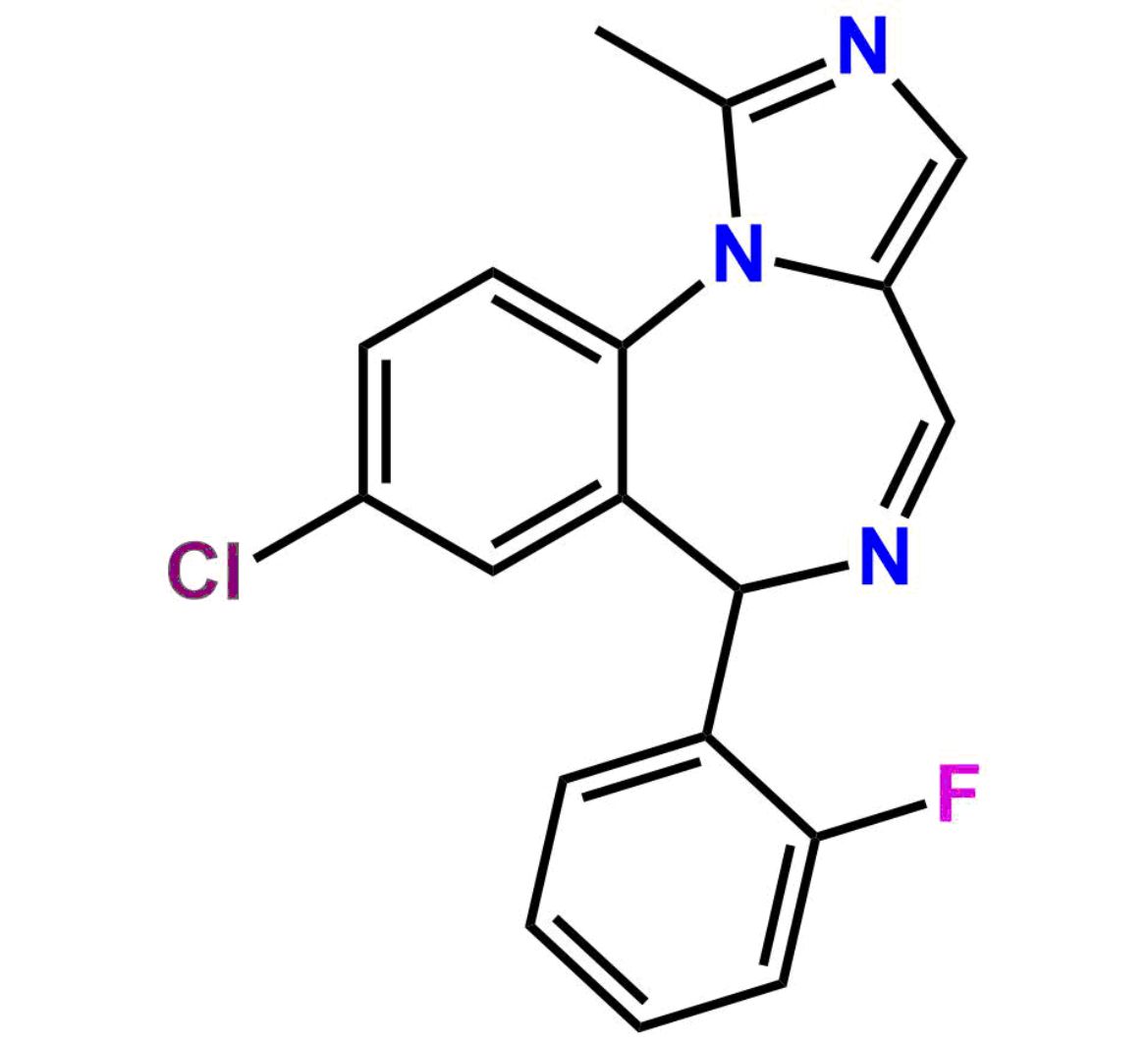
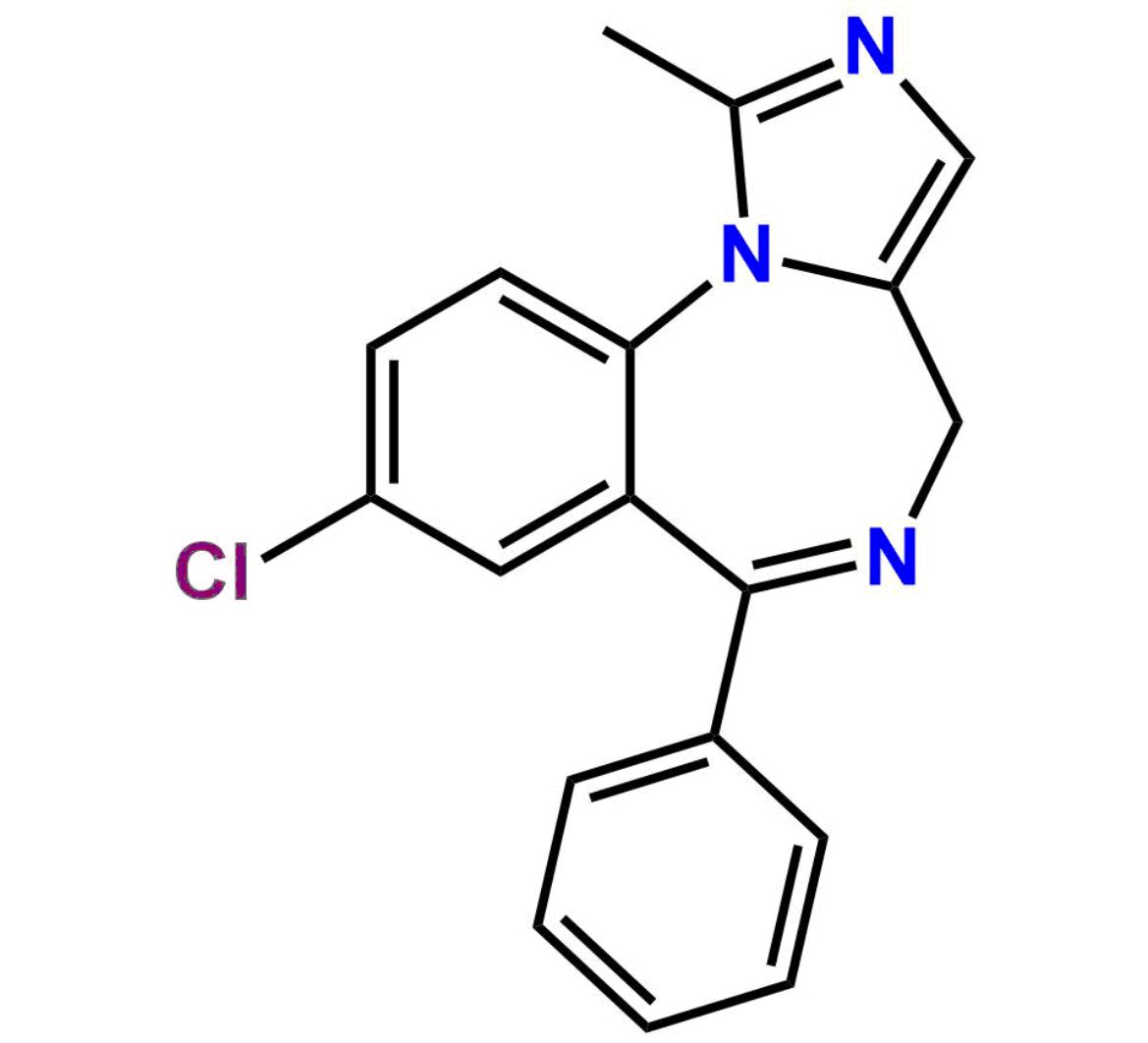
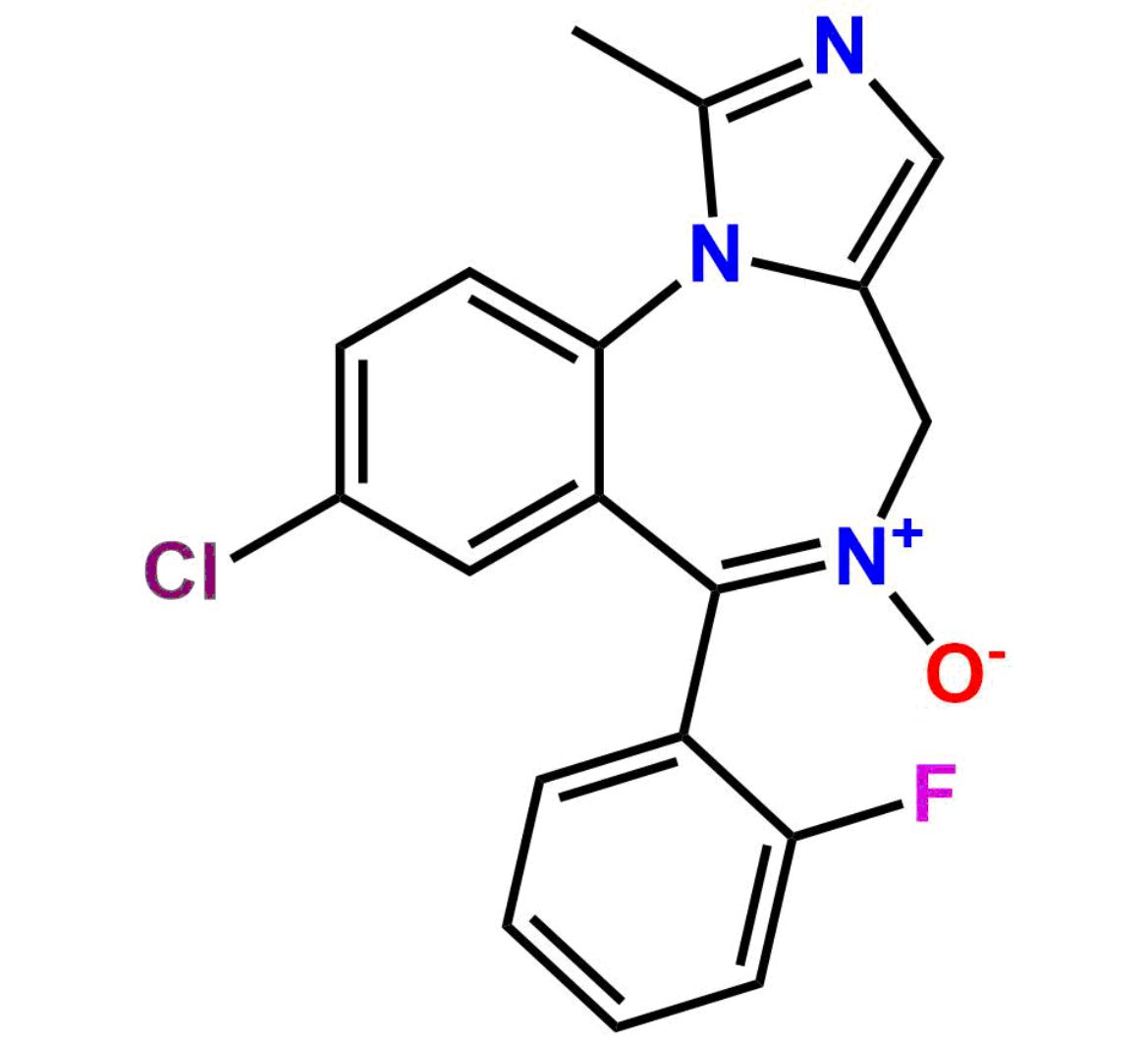
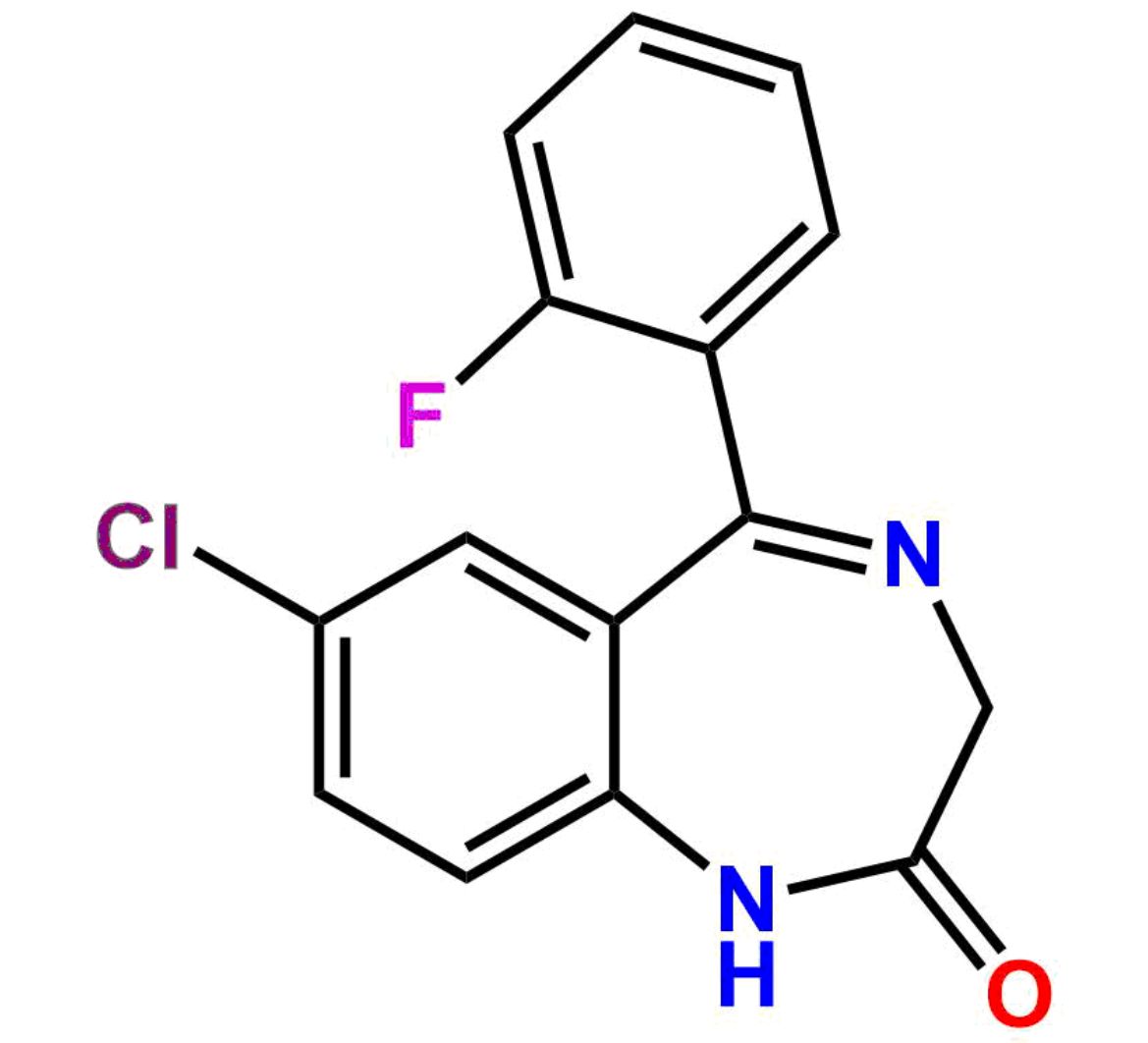
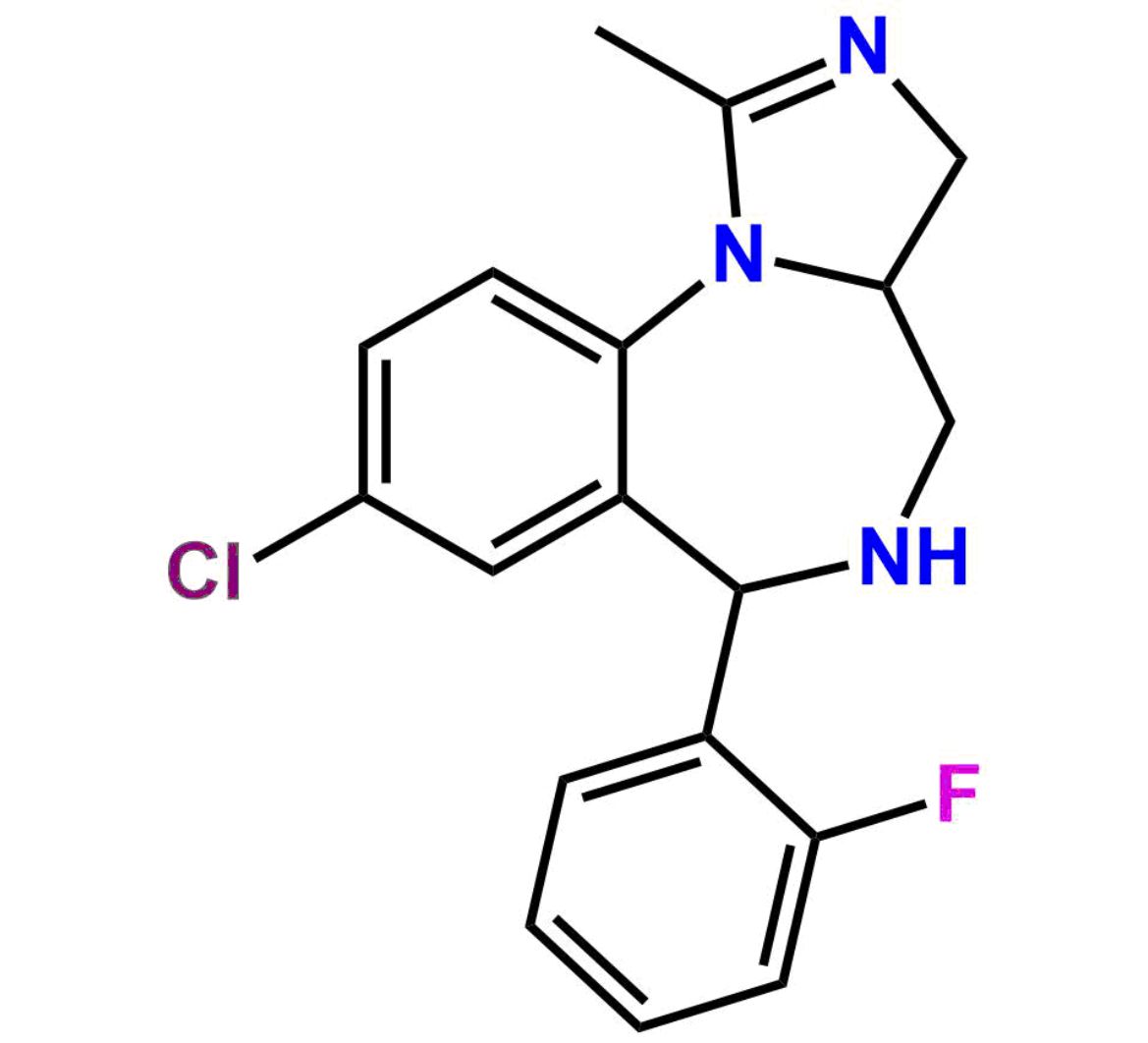
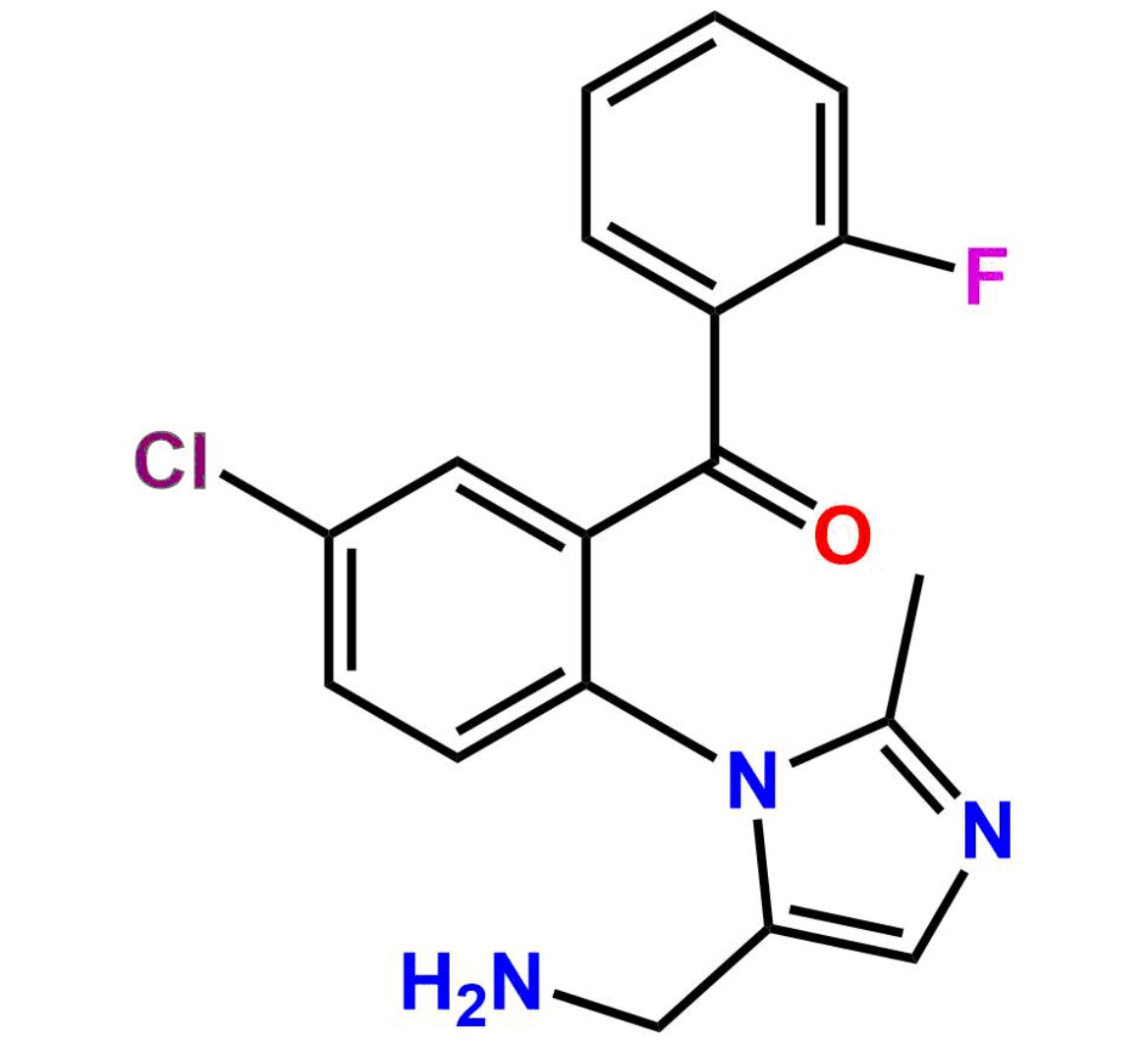
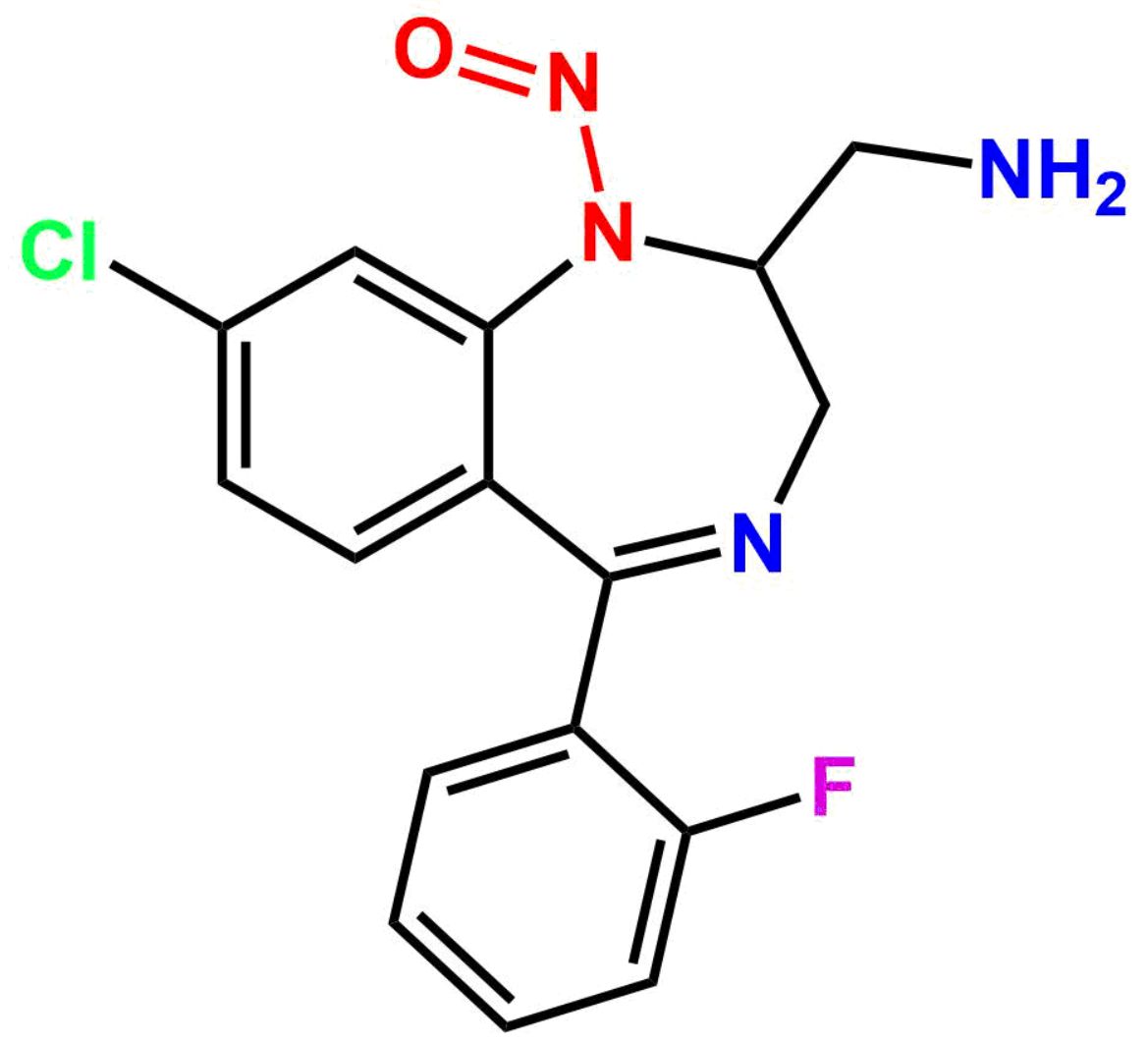
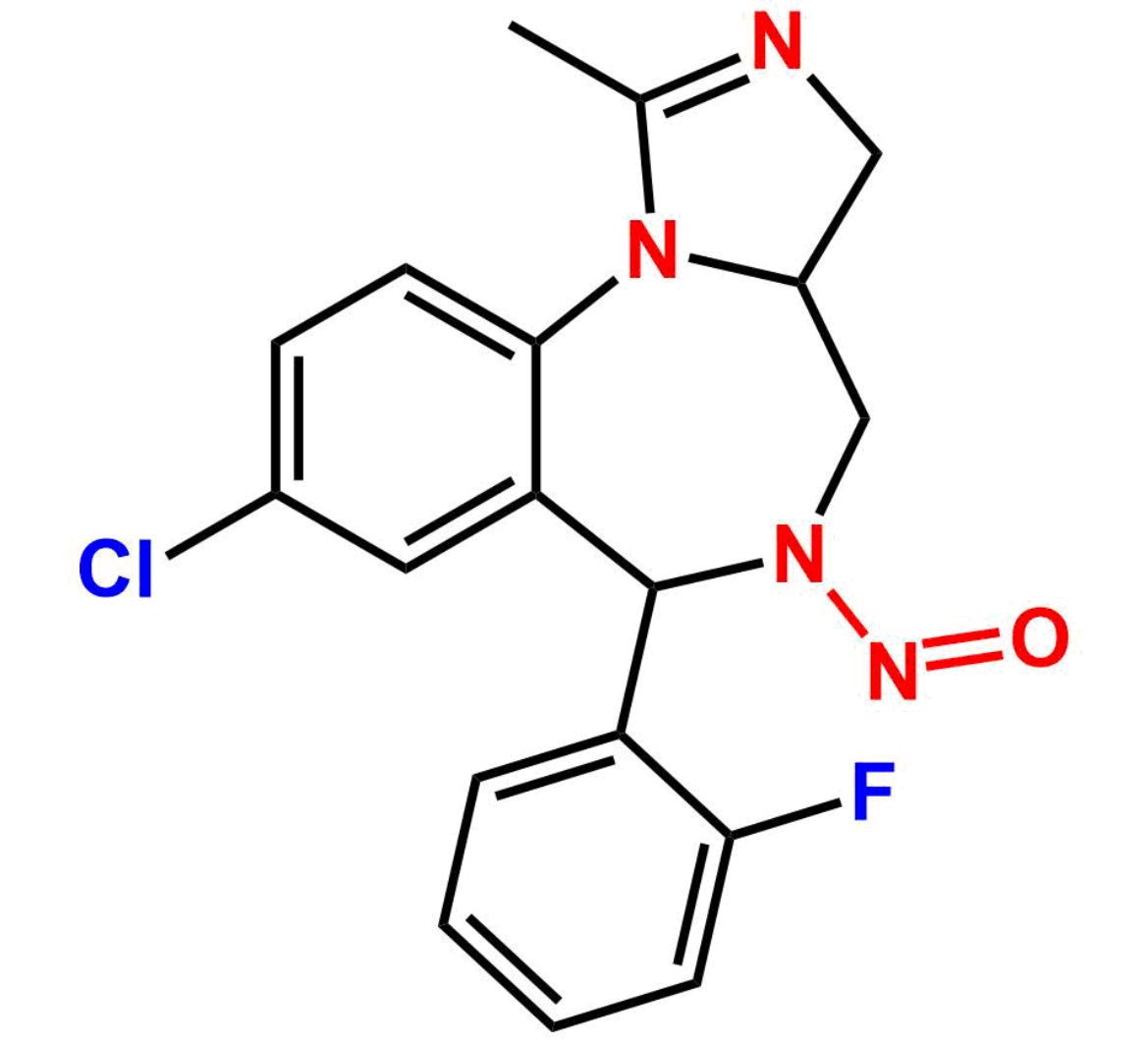
This impurity is related to Midazolam Impurity 13 is provided with comprehensive characterization data in accordance with regulatory guidelines. It is suitable for use in analytical method development, method validation (AMV), Quality Control (QC) applications for Abbreviated New Drug Applications (ANDA) and commercial production of Asenapine.
Midazolam Impurity 13 is used as a reference standard in analytical research. It ensures consistency of formulations.
Chemical Name:6-chloro-2-(chloromethyl)-4-(2-fluorophenyl)quinazoline 3-oxideCountry of Origin: India Product Category: Impurity Reference StandardAPI NAME: Midazolam Molecular Formula: C15H9Cl2FN2O
Molecular Weight: 323.1
Storage: Store in a cool, dry place.